


TLR 7/8 AS AN IMMUNOTHERAPY TARGET
Antigen-presenting cells (APCs) play a critical role in initiating the cancer-immunity cycle1
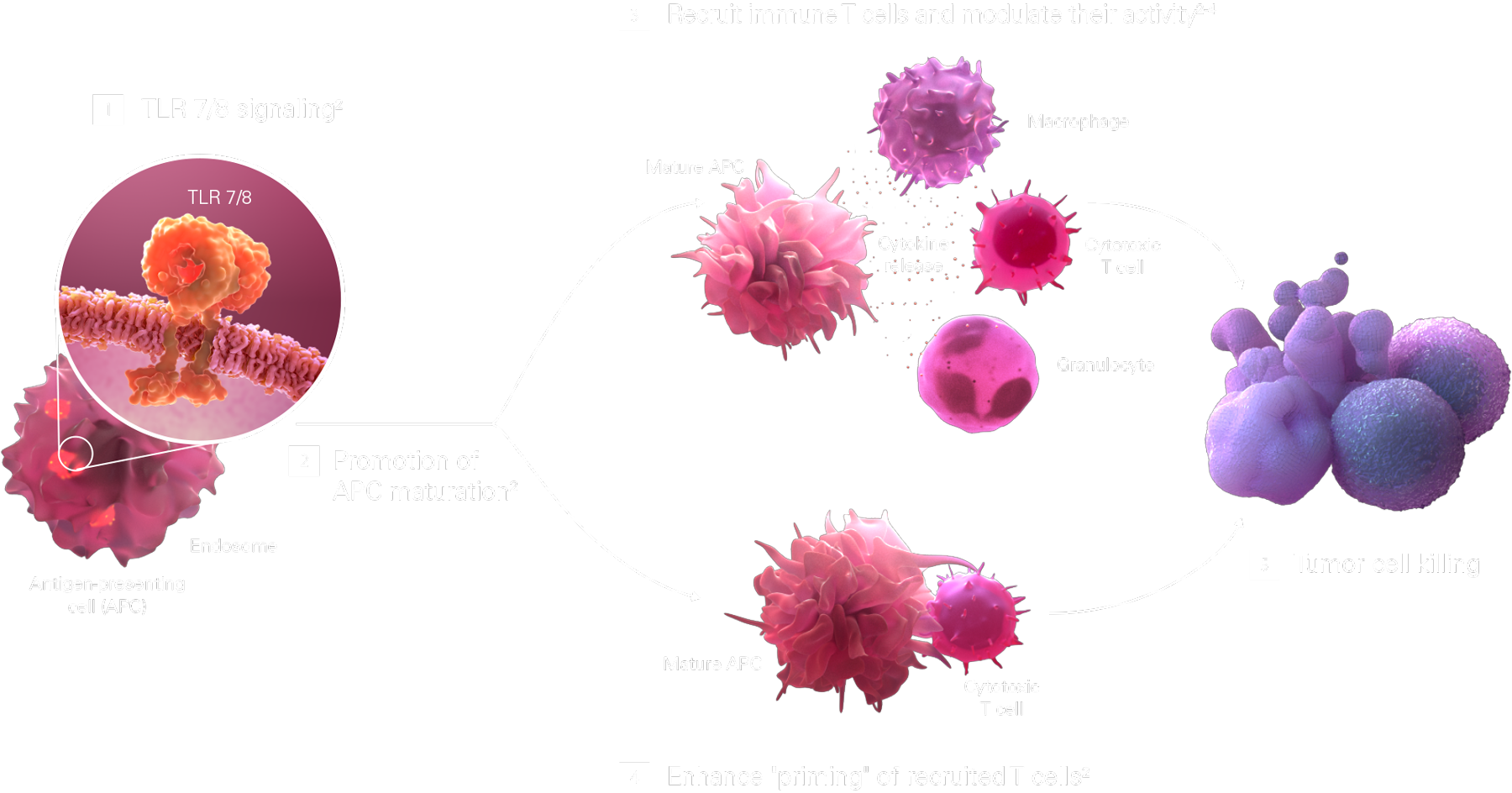
The toll-like receptor (TLR) 7/8 pathway activates APCs that prime T cells and recruit immune cells within the tumor microenvironment2,4
Activation of the TLR 7/8 signaling pathway [1] promotes the maturation of APCs, [2] which both:
TLR 7/8 agonists could improve antitumor immune response when combined with other immunotherapies2,4
Nektar is exploring sustained and targeted TLR 7/8 signaling in combination with other immunotherapies5
References: 1. Chen DS, Mellman I. Oncology meets immunology: the cancer-immunity cycle. Immunity. 2013;39(1):1-10. 2. Adams S. Toll-like receptor agonists in cancer therapy. Immunotherapy. 2009;1(6):949-964. 3. Rabeony H, Pohin M, Vasseur P, et al. IMQ-induced skin inflammation in mice is dependent on IL-1R1 and MyD88 signaling but independent of the NLRP3 inflammasome. Eur J Immunol. 2015;45:2847-2857. doi:10.1002/eji.201445215. 4. Singh M, Khong H, Dai Z, et al. Effective innate and adaptive antimelanoma immunity through localized TLR7/8 activation. J Immunol. 2014;193:1-10. doi:10.4049/jimmunol.1401160. 5. Kivimäe S, Pena R, Hennessy M, et al. Systemic anti-tumor immunity and immune memory formation by a novel TLR7/8 targeting agent NKTR-262 combined with CD122-biased immunostimulatory cytokine NKTR-214. Presented at: Society for Immunotherapy of Cancer annual meeting. November 7-11, 2018; Washington, D.C., USA.
ENGAGING IMMUNE RESPONSE THROUGH THE IL-15 PATHWAY
Both innate and adaptive immunity are needed for sustained immune response
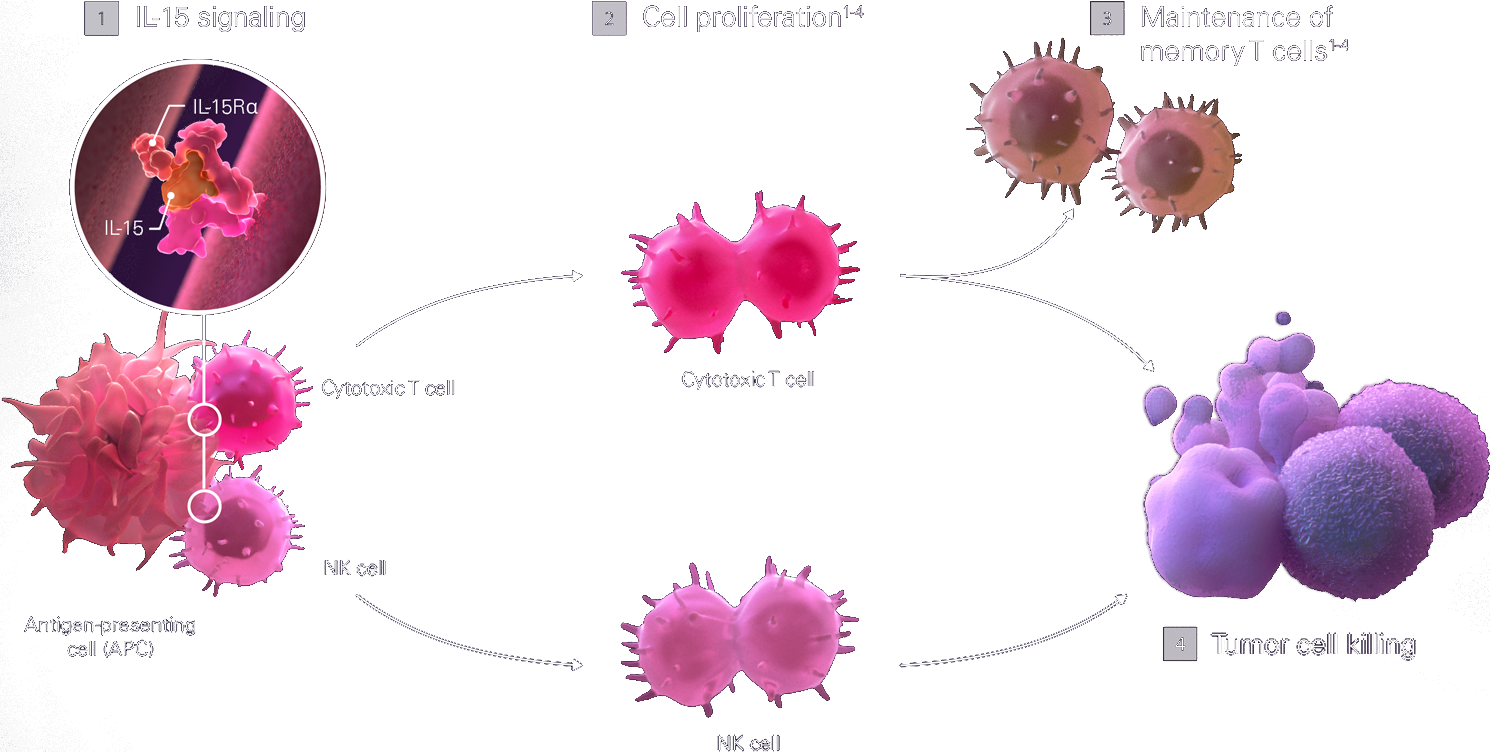
Activation of the IL-15 pathway engages both adaptive and innate antitumor immunity
IL-15 signaling [1] induces both the proliferation and activation [2] of both NK cells and of cytotoxic T cells to1,3,4
IL-15 agonists show potential in combination with other oncology therapeutics1
Nektar is investigating sustained and controlled engagement of the IL-15 pathway6
References: 1. Robinson TO, Schluns KS. The potential and promise of IL-15 in immuno-oncogenic therapies. Immunol Lett. 2017;190:159-168. 2. Messaoudene M, Frazao A, Gavlovsky PJ, Toubert A, Dulphy N, Caignard A. Patient's natural killer cells in the era of targeted therapies: role for tumor killers. Front Immunol. 2017;8:683. doi:10.3389/fimmu.2017.00683. 3. Osborne LC, Abraham N. Regulation of memory T cells by yc cytokines. Cytokine. 2010;50(2):105-113. 4. Kennedy MK, Glaccum M, Brown SN, et al. Reversible defects in natural killer and memory CD8 T cell lineages in interleukin 15-deficient mice. J Exp Med. 2000;191(5):771-780. 5. Stonier SW and Schluns KS. Trans-presentation: a novel mechanism regulating IL-15 delivery and responses. Immunol Lett. 2010;127(2):85-92. doi:10.1016/j.imlet.2009.09.009. 6. Data on File. Nektar Therapeutics.
THE ROLE OF THE IL-2 PATHWAY IN IMMUNE RESPONSE
Higher levels of cytotoxic T cells and natural killer (NK) cells raise tumor cell killing potential in the tumor microenvironment1,2
The IL-2 pathway can both stimulate and regulate immune responses
TAKE A CLOSER LOOK - CLICK BELOW TO WATCH THE IL-2 PATHWAY VIDEO
IMMUNE SYSTEM REGULATION:
IL-2 cytokine binding of the trimeric complex [alpha (CD25) + beta (CD122) + gamma (CD132)] on Tregs [2] causes their activation and proliferation, and thereby immune regulation [4]7-10
IMMUNE SYSTEM STIMULATION:
IL-2 cytokine binding of the dimeric complex [beta (CD122) + gamma (CD132)] on cytotoxic T cells and NK cells [2] induces their proliferation and activation leading to immune activation [3]7
Preferential dimeric activation of the IL-2 receptor is a target for further research7
References: 1. Chen DS, Mellman I. Oncology meets immunology: the cancer-immunity cycle. Immunity. 2013;39(1):1-10. 2. Messaoudene M, Frazao A, Gavlovsky PJ, et al. Patient's natural killer cells in the era of targeted therapies: role for tumor killers. Front Immunol. 2017;8:683. doi:10.3389/fimmu.2017.00683. 3. Tumeh PC, Harview CL, Yearley JH, et al. PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature. 2014;515:568-583. 4. Halle S, Keyser KA, Stahl FR, et al. In vivo killing capacity of cytotoxic t cells is limited and involves dynamic interactions and t cell cooperativity. Immunity. 2016;44(2):233-245. 5. Cursons J, Souza-Fonseca-Guimaraes F, Foroutan M. et al. A gene signature predicting natural killer cell infiltration and improved survival in melanoma patients. Cancer Immunology Research. 2019;7(7):1162-1174. 6. Boyman O, Sprent J. The role of interleukin-2 during homeostasis and activation of the immune system. Nature Rev Immunol. 2012;12(3):180-190. 7. Charych D, Khalili S, Dixit V, et al. Modeling the receptor pharmacology, pharmacokinetics, and pharmacodynamics of NKTR-214, a kinetically-controlled interleukin-2 (IL2) receptor agonist for cancer immunotherapy. PLoS One. 2017;12(7):e0179431. doi:10.1371/journal.pone.0179431. eCollection 2017. 8. Ye C, Brand D, Zheng SG. Targeting IL-2: an unexpected effect in treating immunological diseases. Signal Transduct Target Ther. 2018;3:1-10. 9. Johnson K, Choi Y, Wu Z, et al. Soluble IL-2 receptor beta and gamma subunits: ligand binding and cooperativity. Eur Cytokine Netw. 1994;5:23-34. 10. Liparoto SF, Myszka DG, Wu Z, et al. Analysis of the role of the interleukin-2 receptor gamma chain in ligand binding. Biochemistry. 2002;41:2543-2551.